We get asked all the time about how to measure the pH of skincare products – what pH is, when you need to measure it and how to measure it. Here you’ll discover the answers to these questions, as well as why we measure it and how to adjust your pH testing equipment.
What is pH?
pH is a measure on a scale of 0-14 of how acidic or alkaline something is.
A product with a pH of 7 is neutral, a pH of less than 7 is acidic, and a pH greater than 7 is alkaline. pH is only measurable where water is present, as it indicates the concentration of hydrogen ions.

Our skin has a specific pH value, which normally averages around pH 5, and varies from one body part to another and from one person to another. It even changes during the aging process. This acidic nature plays a vital role in maintaining healthy skin.
When do you need to measure the pH of skincare products?
It is common practice to measure the pH of cosmetic products that contain water, such as skincare (creams, lotions, milks, cleansers), body care (body washes, body scrubs) and haircare preparations (shampoo, conditioner, hair masks).

There is no need to test anhydrous products like body butter, sugar scrubs, salves and lip balm as they do not contain water.
What pH should skincare products be?
We get asked this question A LOT! Some people are convinced that skincare products should be a neutral pH (pH 7). Others think that skincare products should have the same pH as our skin (pH 4.5-5.5).
The skin’s natural pH is slightly acidic (pH 4.5-5.5), so it is a good target pH for your products, too.
However, the correct pH for your natural skincare products is determined by the ingredients in your product formula. In particular, if you are using natural preservatives, you will need to adjust the pH of your product so that it is within the optimal range for your preservative to be effective.
Some people worry about using a product that isn’t within the pH range of our skin. Our skin actually has a mechanism of balancing its pH; even if you wash your face with a product that has a pH of 8, the skin’s natural pH levels will be restored over time.
Why do you need to measure and adjust the pH of skincare products?
The pH of natural skincare products is very important if you are using any pH-sensitive ingredients, like naturally derived preservatives or certain active ingredients.
Almost all of the naturally derived preservatives (sodium benzoate, potassium sorbate and salicylic acid, for example) have very strict pH requirements to ensure they work properly, which means measuring (and adjusting) the pH is necessary when protecting your product with natural preservatives.
Preservatives active at a lower pH can lose their efficiency quite quickly when the pH increases; for instance potassium sorbate is active at about 70% at pH 4.5 and at about 35% at pH 5. Most natural preservatives require a pH below 5.
If you are using certain active ingredients, you will need to be aware of the pH of your product. AHAs (alpha hydroxy acids) need pH 4 or lower to be active as chemical exfoliants. Ascorbic acid (the common form of Vitamin C), requires a pH below 3.5 to be active. Niacinamide (Vitamin B3) is most stable at pH 6. Always check with your supplier of active ingredients what pH levels are needed for a specific ingredient.
How do you measure the pH of skincare products?
The two options for measuring pH are pH strips and a pH meter, and there are pros and cons to each.
Measuring pH using pH strips

PH strips are small strips with a pH-sensitive area on one end that will change color based on the pH of the substance that it is exposed to.
This color is then compared to a color chart to obtain a pH reading. These strips are fairly inexpensive and very easy to use.
NOTE: Just make sure the pH strips you purchase have a wide range like can test pH levels from 0-14.
Another con to using pH strips is that you’ll likely need to use more than one strip per product, each time you adjust your formula, you’ll likely need a new pH reading.
Meaning that for each product, you’ll probably go through several strips.
|
Advantages |
Disadvantages |
|
Inexpensive |
Not very precise, only provides a rough estimate |
|
Simple to use |
Several consequent measurements are needed to adjust the pH, so you may end up using many pH strips for one product |
|
Easily accessible |
|
|
Can be used for all product types including emulsions |
Measuring pH using a pH meter
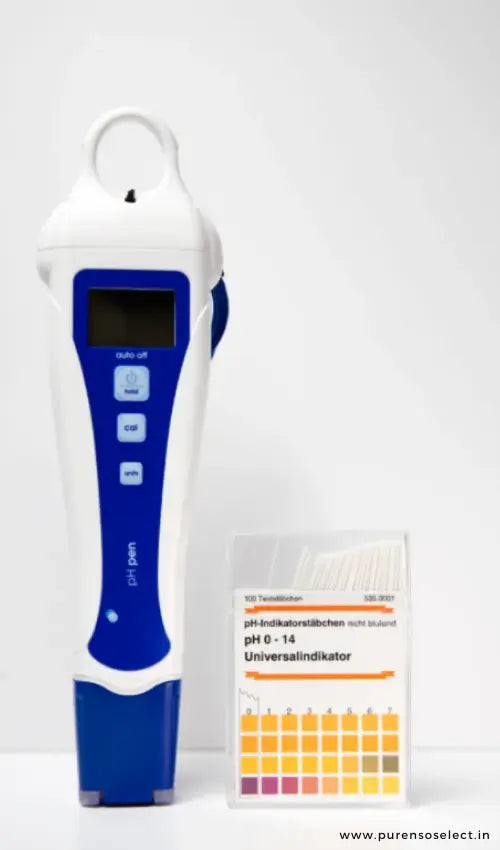
These meters can be pricey, but they can measure the exact pH of the product you’re making.
PH meters are a good option for anyone who is making products for others and needs precise measurements.
While you’ll only need one meter to read the pH of a product multiple times, they need proper care and storage along with precise calibration.
A pH meter may also need an emulsion probe to read pH levels on those products.
What tool you use to measure your products’ pH is ultimately up to you and what works best for your products!
|
Advantages |
Disadvantages |
|
Gives precise measurements (to one decimal point) |
Expensive |
|
Needs regular cleaning and proper storage |
|
|
Requires calibration before use |
|
|
An additional emulsion probe may be needed |
For beginners, pH strips can be a good choice, but when you want to become more professional you’ll need to learn how to use a pH meter.
If you are using natural preservatives that require a precise pH in order to be effective then it’s advisable to use a pH meter to test and adjust your pH.
If you plan to sell your natural skincare products and are sending products to a lab for microbiological testing then you’ll definitely need to measure and adjust the pH of your products very precisely, which requires a pH meter.
How to Measure pH in Skincare Recipes
You will need a sample of your product added to a sanitized glass beaker or container. (sample of around 1-5 grams)
- Place your sample in a glass beaker
- Dilute with distilled water at a ratio of 10% product 90% water (note: some products like toners may not need to be diluted)
- Stir well then test your pH
(There are two fairly simple ways to test the pH in skincare recipes. pH strips and pH meters. Both have their pros and cons but are fairly simple to use!)
How to adjust the pH of your natural skincare products
Testing pH is just the first step; the next is to adjust it. The steps you need to follow are:
- Create an appropriate pH adjustment solution to either increase or decrease the pH. (If you use sodium hydroxide you need to do this with a lot of care as it is extremely caustic.)
- Record the amount of the adjustment solution that you used to adjust the pH to the desired measurement.
- After you have adjusted the pH of your product, calculate the amount of solid/pure pH adjuster used this is different to the amount of pH adjustment solution).
- Rewrite your formula to include the pH adjuster.
How to Increase pH Levels

How to Decrease pH Levels

In order to decrease pH, you can use citric acid or lactic acid. Citric acid has a pH between 3 and 6 and can be found in powder or liquid form.
Adding small amounts of citric acid to a formulation can reduce pH levels; it is also a chelator and can bind any metal compound in the formulation to increase your preservative efficacy.
NOTE: You will need to mix together 10% citric acid and 90% distilled water before adding to your product. A good starting place would be 1 gram of citric acid + 9 grams distilled water.
Once your mixture is combined, you will simply add just a few drops to your product formulation and mix well.
Test the pH once again after adding to make sure it's at a suitable level around 5-5.5.
Lactic acid can be harder to come by than citric acid. It is typically sold as a liquid with a maximum concentration of 90% like this one.
Please keep in mind that the pH of Lactic acid is 2.43, making it very acidic.
At a high concentration, it can be very corrosive and should be worked with very carefully!
You will need to dilute it further with distilled water before adding it to your formulations. You can dilute it to 10%, 25% or 40%.
When used properly in a formulation, Lactic acid can be very moisturizing and work very well with your preservatives!
When you’re just starting out making your own skincare products or trying something new, it is imperative to remember to work in small batches at first and be prepared to start over!
Work in small increments when adjusting pH; you can always add more pH adjusting compounds if you didn’t add enough!
You should also measure the pH often, after each addition of any pH adjusting compound.
If you adjust your pH and it becomes too low, you risk ingredients in your formula degrading; so when adjusting pH, start low and go slow!
Do not use any home products such as regular baking soda, lemon juice, or vinegar to adjust your products’ pH!



Comment
Thankyou for this post. It helped me.